News & Review: AI Predicts Single‑Cell Spatial Transcriptomes from Histology Images
- Eghosa Arovo

- Jan 7
- 3 min read
Can a standard H&E image reveal the full spatial expression of genes at single‑cell resolution? New research reviewed here suggests it might. Discover what this means for histology‑based studies.

Introduction
In this post, we’re reviewing a recent scientific development reported on EurekAlert!, a trusted news platform from the American Association for the Advancement of Science (AAAS). The featured study introduces a deep learning model called PRTS (Pathology-driven Reconstruction of Transcriptomic States), which predicts single-cell spatial transcriptomic maps directly from H&E-stained histology images.
While the technology is still in early stages, its implications for histology-based research are significant—especially for those working with tumour tissues, complex tissue architecture, or large-scale datasets where spatial transcriptomics (ST) may not be accessible.
What the Study Shows
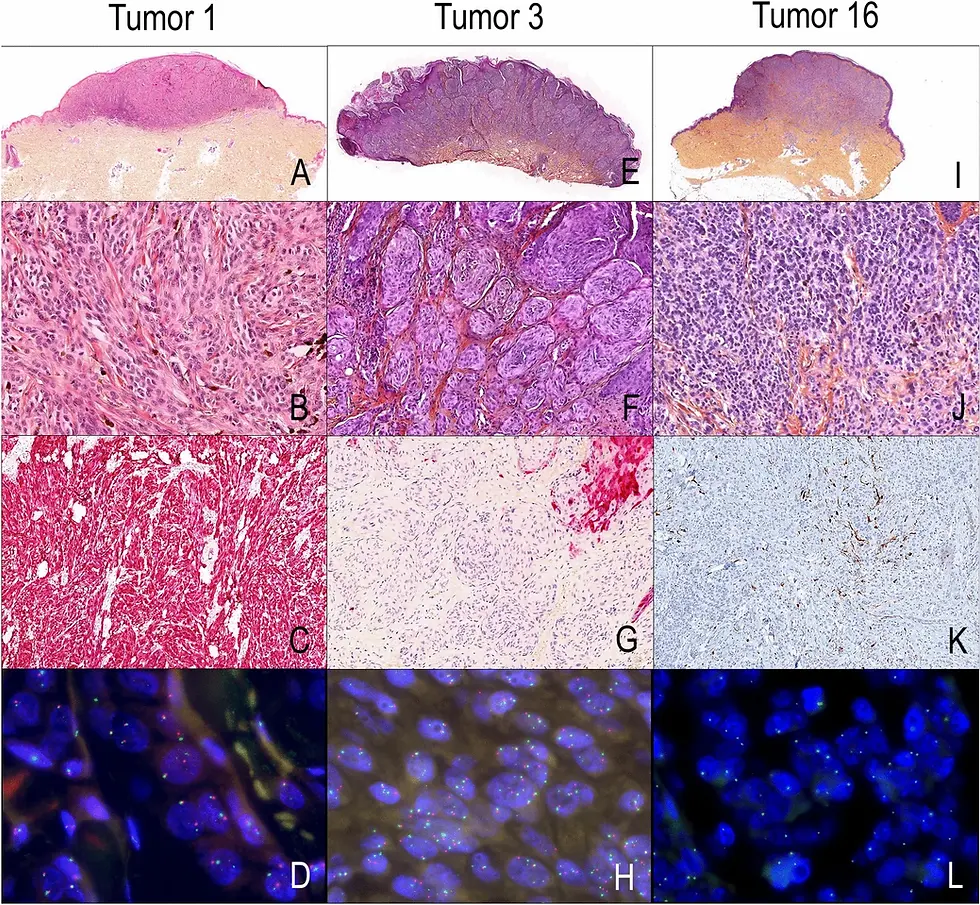
PRTS was developed by researchers led by Professor Fei Ling at South China University of Technology. According to the report, the model takes standard H&E-stained images and predicts gene expression at single-cell resolution—achieving up to 27 times higher spatial resolution than traditional ST spot-based methods.
The team tested this model on mouse brain, human lung, and breast cancer tissues. It was able to accurately infer spatial gene expression patterns and distinguish 21 unique cell subtypes, aligning closely with data from experimental ST platforms. These predictions not only matched gene-level resolution but also recapitulated known spatial arrangements of cells within the tissue.
This opens up new possibilities for tissue studies that combine traditional histology with cutting-edge machine learning. Full details of the study can be found via the EurekAlert! news release.
Why This Matters to Histology Researchers
While the study is focused on computational modelling, it brings histology into sharper focus. The ability of PRTS to generate spatial transcriptomic maps from histology images alone underscores just how much valuable information is embedded in tissue architecture.
For researchers, this reinforces the importance of high-quality H&E staining and slide preparation. The effectiveness of models like PRTS depends heavily on the resolution, clarity, and consistency of histological images. In other words, well-prepared tissue sections don’t just support morphological analysis—they enable computational exploration at the molecular level.
Furthermore, this model bridges a long-standing gap between morphology and molecular biology. Traditionally, histology reveals structure, while transcriptomics reveals gene activity. PRTS attempts to connect these two domains using image-based inference—highlighting the expanding role of histology as a data-rich foundation for integrated research.
Limitations and Considerations
As promising as this development is, it's important to interpret it within context. EurekAlert! is a science communication platform, and it clearly states that it does not independently verify the claims made in the studies it reports. The original research is peer-reviewed, but its real-world utility depends on further validation, replication, and technological accessibility.
It’s also worth noting that PRTS is an inference tool—it doesn’t measure gene expression directly. Instead, it predicts likely patterns based on existing correlations between histology and molecular data. For researchers, this means such tools are best used in complement to experimental approaches, not as replacements.
The Growing Role of Histology in AI & Omics Integration
As AI and machine learning become more embedded in biomedical research, high-quality histology is more important than ever. Histological slides are no longer just a visual aid—they are data inputs for advanced computational models. This means that everything from sectioning and staining to image resolution directly affects the quality of insights that can be extracted.
Whether you're studying cancer, developmental biology, or regenerative medicine, combining traditional histology with digital analysis opens new avenues for understanding complex tissues.
LabNexus Supports Research-Grade Histology
At LabNexus, we’re proud to support researchers working at the intersection of biology, imaging, and computation. Our lab provides high-standard, reproducible histology services for research use only—including H&E staining, special stains, immunohistochemistry (IHC), and high-resolution slide scanning.
With affordable pricing, quick turnaround times, and a strict research-only focus (no diagnostics), LabNexus enables researchers to generate clean, consistent data for image analysis, quantification, and even AI-driven pipelines.
If you’re planning a project that requires histological input—or want to integrate high-quality tissue imaging into your research workflow—we’re here to help.
Book a free consultation today at www.labnexus.co.uk/book-consultation.
References
“PRTS: Predicting Single‑Cell Spatial Transcriptomic Maps from Histological Images.” EurekAlert!, 21 Dec 2025. https://www.eurekalert.org/news-releases/1110775
Junqueira, L.C., Bignolas, G., & Brentani, R.R. (1979). Picrosirius staining plus polarization microscopy for collagen visualisation. Histochem J, 11(4), 447–455.
Rittié, L. (2017). Method for picrosirius red-polarization detection of collagen fibers. Methods Mol Biol, 1627, 395–407.
Bancroft, J.D., & Gamble, M. (2020). Theory and Practice of Histological Techniques (8th ed.). Elsevier.
EurekAlert! Guidelines and Use Policy: https://www.eurekalert.org/releaseguidelines
.png)


Comments