Tissue Microarrays (TMAs): What They Are and Why They’re Powerful for Histology Research
- Eghosa Arovo

- Feb 23
- 4 min read
TMAs let you analyse dozens of tissue samples on one slide — here’s how they work and why researchers use them.

Introduction
In histology and cancer research, one of the biggest challenges is analysing large numbers of tissue samples in a consistent, efficient, and cost-effective way. This is especially true in biomarker studies, cohort comparisons, and translational research.
This is where tissue microarrays (TMAs) become incredibly valuable.
If you’ve never come across TMAs before, they might sound complex. In reality, they are a simple but powerful concept that allows researchers to place many different tissue samples into a single paraffin block — so that all samples can be stained and analysed together under identical conditions.
In this blog, we explain what TMAs are, how they are made, and why they are such a powerful tool for modern histology research.
What is a tissue microarray (TMA)?
A tissue microarray (TMA) is a paraffin block that contains multiple small tissue cores taken from different donor blocks and re-embedded into a single “recipient” (blank) paraffin block.
Each core represents a specific tissue sample — for example:
Different patients’ tumour samples
Treated vs untreated tissue
Control vs disease tissue
Different regions of the same tumour
When thin sections are cut from the TMA block, each microscope slide contains dozens or even hundreds of tissue samples arranged in a grid-like pattern.(Kononen et al., 1998)
How are TMAs made?
The basic workflow for creating a TMA looks like this:
Selection of donor blocksExisting paraffin blocks containing tissue of interest are reviewed. Specific regions (e.g. tumour-rich areas) are marked on H&E-stained slides.
Core extractionA small cylindrical core of tissue (typically 0.6–2.0 mm in diameter) is punched out from each donor block at the selected region.
Insertion into a recipient blockEach tissue core is inserted into a predefined position in a blank paraffin “recipient” block, creating a grid of samples.
Sectioning and stainingThin sections are cut from the completed TMA block and used for H&E, IHC, multiplex IHC, or special stains.
At LabNexus, we can construct custom TMA blocks from your existing paraffin blocks, inserting any tissue of interest into a blank recipient block in a layout designed specifically for your research project.
Why use TMAs in histology research?
TMAs are widely used in cancer and tissue research because they offer several major advantages.
1. High-throughput analysis
With TMAs, you can analyse many samples at once:
20, 50, 100, or even 300+ tissue cores on one slide
All samples stained under identical conditions
All samples imaged and analysed in a single workflow
This is ideal for biomarker screening, cohort studies, and comparative analysis.(Kononen et al., 1998)
2. Excellent consistency and reproducibility
Because all tissue cores are stained on the same slide:
Antibody concentration is identical for all samples
Incubation times are identical
Chromogen development is identical
This greatly reduces technical variation and makes quantitative comparison between samples far more reliable than using serial full sections.
3. Major savings in tissue, reagents, and cost
TMAs dramatically reduce resource use:
Far fewer slides need to be stained
Far less antibody and chromogen is consumed
Far less technician time is required
This makes TMAs especially cost-effective for large cohort studies and multiplex IHC projects.
4. Perfect match for multiplex IHC
TMAs and multiplex staining are a natural pairing.
By applying multiplex chromogenic IHC or multiplex IF to TMA sections, you can:
Profile multiple biomarkers across large cohorts
Generate spatial biomarker datasets
Identify expression patterns linked to outcome or phenotype
This is now a standard approach in translational cancer research.(Ramos-Vara & Miller, 2014)
What kinds of projects benefit most from TMAs?
TMAs are especially useful for:
Cancer biomarker discovery and validation
Translational and retrospective cohort studies
Drug response and treatment-stratification research
Immune profiling of tumour microenvironments
Comparing multiple disease subtypes
Control vs disease tissue comparisons
They are also widely used for antibody validation and quality control in histology labs.
Important limitations to be aware of
Although TMAs are extremely powerful, they are not perfect for every project.
Key limitations include:
Sampling biasEach core represents only a small region of tissue. Heterogeneous tumours may require multiple cores per case.
Loss of large-scale architectureTMAs are not ideal for studying whole-tumour morphology or invasion fronts.
Core loss during sectioningSome tissue types may detach during cutting or staining.
These limitations are usually managed by careful core selection and using multiple cores per case when needed.(Kononen et al., 1998)
TMAs at LabNexus
At LabNexus, we support TMA-based research from start to finish.
We offer:
Custom TMA block construction from your existing paraffin blocks
Insertion of any tissue of interest into a blank recipient block
Flexible layouts tailored to your study design
Sectioning of TMA blocks
Single-plex and multiplex chromogenic IHC
Optional slide scanning and image analysis support
This allows you to transform a collection of individual tissue blocks into a high-throughput, multiplex-ready research platform.
Please note: LabNexus provides histology services for research purposes only. We do not process diagnostic samples.
Conclusion
Tissue microarrays are one of the most powerful tools in modern histology research.
They allow researchers to:
Analyse large numbers of tissue samples efficiently
Preserve valuable tissue
Improve reproducibility
Reduce cost and reagent use
Combine seamlessly with multiplex IHC
Whether you are planning a biomarker study, a cancer cohort project, or a high-throughput multiplex experiment, TMAs can dramatically elevate the scale and quality of your histology research.
At LabNexus, we can help you build custom TMA blocks from your existing tissue and run high-quality histology and IHC workflows on them — quickly, reliably, and affordably.
References
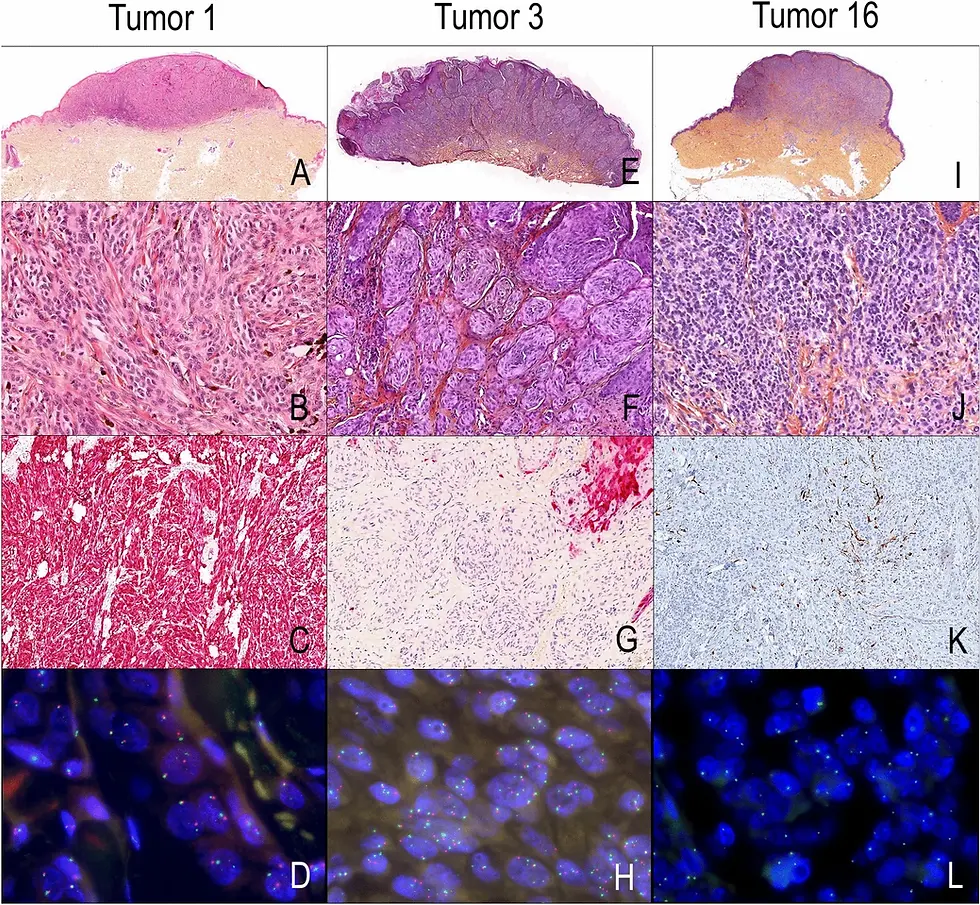
"Figure 2": Nature: https://www.nature.com/articles/s41598-021-81352-y
Kononen, J. et al. (1998). Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nature Medicine, 4, 844–847.
Ramos-Vara, J.A., & Miller, M.A. (2014). When tissue antigens and antibodies get along: Revisiting the technical aspects of immunohistochemistry. Veterinary Pathology, 51(1), 42–87.
Bancroft, J.D., & Gamble, M. (2020). Theory and Practice of Histological Techniques (8th ed.). Elsevier.
Hewitt, S.M. (2004). Design, construction, and use of tissue microarrays. Methods in Molecular Biology, 264, 61–72.
.png)

Comments